Search
High-flow nasal oxygen (HFNO) is frequently used in hospitals, producing droplets and aerosols that could transmit SARS-CoV-2. Our aim was to determine if a headbox could reduce droplet and aerosol transmission from patients requiring HFNO.
In the absence of an established gold standard, an understanding of the testing cycle from individual exposure to test outcome report is required to guide the correct interpretation of SARS-CoV-2 reverse transcriptase real-time polymerase chain reaction (RT-PCR) results and optimise the testing processes.
The aim of this study was to explore the relationship between emotional health and wellbeing and support needs of perinatal women during the COVID-19 pandemic, and to understand their experiences and need for support. This is a potentially vulnerable group and a critical developmental phase for women and infants.
Public health measures targeting coronavirus disease 2019 have potential to impact transmission of other respiratory viruses. We found 98.0% and 99.4% reductions in respiratory syncytial virus and influenza detections, respectively, in Western Australian children through winter 2020 despite schools reopening. Border closures have likely been important in limiting external introductions.
The emergence of the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), sparking a global pandemic, has driven an imperative to quickly design and conduct treatment studies. We strongly propose a national, coordinated approach for randomised controlled trials (RCTs) for coronavirus disease 2019 (COVID-19), future pandemics and inter-pandemic periods in Australia.
Providing a safe and effective coronavirus disease 2019 (COVID‐19) vaccination program is required to mitigate against the current and future negative impacts on the health and wellbeing of all Australians from COVID‐19. An effective vaccination program is a key element required to facilitate economic recovery, safe movement throughout and beyond Australia and a return to the quality of life previously experienced.
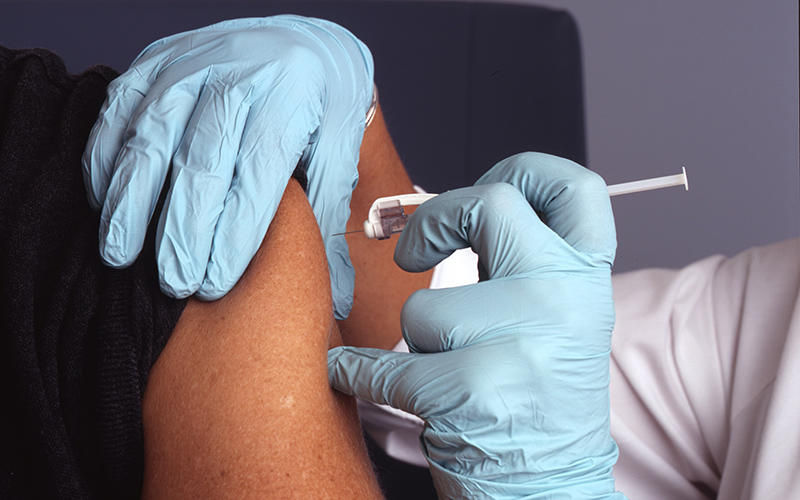
A world-leading international trial examining the immune boosting benefits of the tuberculosis vaccine, BCG, has found it does not protect healthcare workers against COVID-19.

Australia’s TGA has granted a provisional determination to Pfizer, allowing the pharmaceutical company to apply for approval to extend its COVID-19 vaccine to children aged 6 mths - 4 yrs.

Western Australia’s Omicron outbreak is far from over, with new modelling showing the number of total infections is only at its half-way point.

WA’s current Omicron COVID-19 outbreak could jump by 147,000 cases if mask mandates are abandoned before the Easter long weekend, according to sophisticated new modelling.
