Search

We are looking for healthy young adults to take part in a study that will help us learn more about a new pertussis vaccine that we hope will offer greater protection from whooping cough.
The Kids Research Institute Australia is looking for healthy babies (born from 35 weeks of pregnancy onwards) to take part in an important study evaluating a new medication that may help prevent Respiratory Syncytial Virus (RSV) disease.
This is your chance to play a vital role in shaping Australia's COVID-19 vaccine program
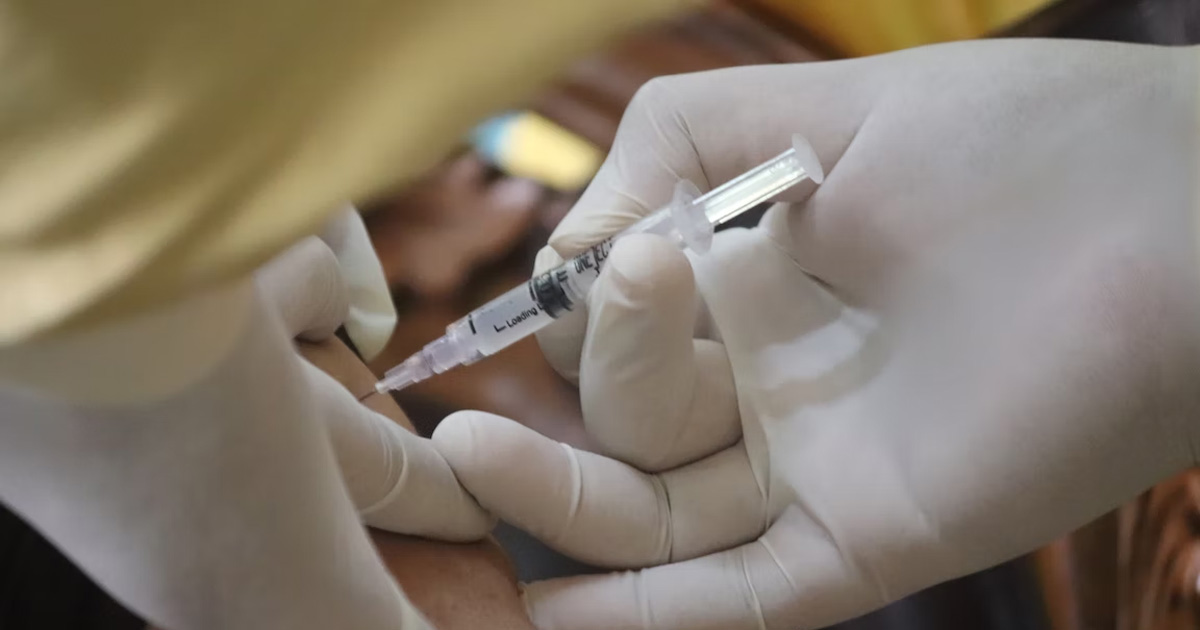
Contact us If you'd like to get in touch, please contact us by phone or email. Phone: 0400 450 240 Email: rhyme@telethonkids.org.au Background The
Allergies have been on the rise in Australia over the past 20 years, with three out of every 10 babies born each year now developing a food-related
The year 2020 has been marked by possibly the greatest health challenge to face modern society. The emergence of COVID-19 as a novel human pathogen
Coming up in 2021 Contact us We have a a study to suit every age range in 2021! From babies at just six weeks for the FluBub Study, through to
Contact us If you'd like to get in touch, please contact us by phone or email. Phone: 0400 450 240 Email: vtg@thekids.org.au Pneumococcal disease is
Contact us If you'd like to get in touch, please contact us by phone or email. Phone: 0400 450 240 Email: vtg@thekids.org.au The Vaccine Trials Group
CHIP Study Update
